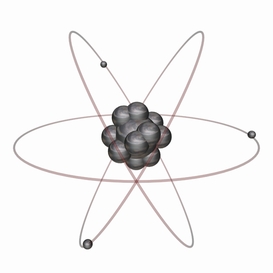
Rutherford (1911)-
-Discovered the nucleus
-Nucleus: small,dense, and positively charged
- A nucleus is in an atom. An atom is mostly empty space
There are two different models. There is. One by Niels Bohr and another by Erwin Schrodinger
Bohr's Planetary Model
- e- In a particular path have a fixed energy level
- Energy level for e- is the region around the nucleus where it is likely to be moving
- The higher the energy level, the farther the e- is from the nucleus. So the lower the energy the closer it gets.
- Lowest energy level = lowest step on periodic table
- e- can change positions or energy level (it. Can move in the atom)
Quantum Mechanical Model
- Erwin Schrodinger described the modern atom based on probability of the location of an electron
- A.K.A The Fuzzy Cloud
- Dense area = high probability
- Quantum of energy is the amount of energy needed to move the electron from its present energy level to the next highest. (Moves farther from nucleus)
- These levels are made of energy sublevels . The sublevels are made of orbitals.
- Energy levels are labeled by principal quantum numbers (n); Range from 1-7
Energy Sublevels
- Energy sublevels are located within each principal quantum number(n)
- There is 4 sublevels : s,p,d,f
- Maximum number of e- that occupy a level n= 2n2
-Discovered the nucleus
-Nucleus: small,dense, and positively charged
- A nucleus is in an atom. An atom is mostly empty space
There are two different models. There is. One by Niels Bohr and another by Erwin Schrodinger
Bohr's Planetary Model
- e- In a particular path have a fixed energy level
- Energy level for e- is the region around the nucleus where it is likely to be moving
- The higher the energy level, the farther the e- is from the nucleus. So the lower the energy the closer it gets.
- Lowest energy level = lowest step on periodic table
- e- can change positions or energy level (it. Can move in the atom)
Quantum Mechanical Model
- Erwin Schrodinger described the modern atom based on probability of the location of an electron
- A.K.A The Fuzzy Cloud
- Dense area = high probability
- Quantum of energy is the amount of energy needed to move the electron from its present energy level to the next highest. (Moves farther from nucleus)
- These levels are made of energy sublevels . The sublevels are made of orbitals.
- Energy levels are labeled by principal quantum numbers (n); Range from 1-7
Energy Sublevels
- Energy sublevels are located within each principal quantum number(n)
- There is 4 sublevels : s,p,d,f
- Maximum number of e- that occupy a level n= 2n2
Citations:
- Westin, Brian. "Detailed Animated Atom 3D Model." Flickr. Yahoo!, 03 Jan. 2012. Web. 29 May 2014.
- Bozeman Science. "Quantum Mechanical Model." YouTube. YouTube, 06 Aug. 2013. Web. 29 May 2014.