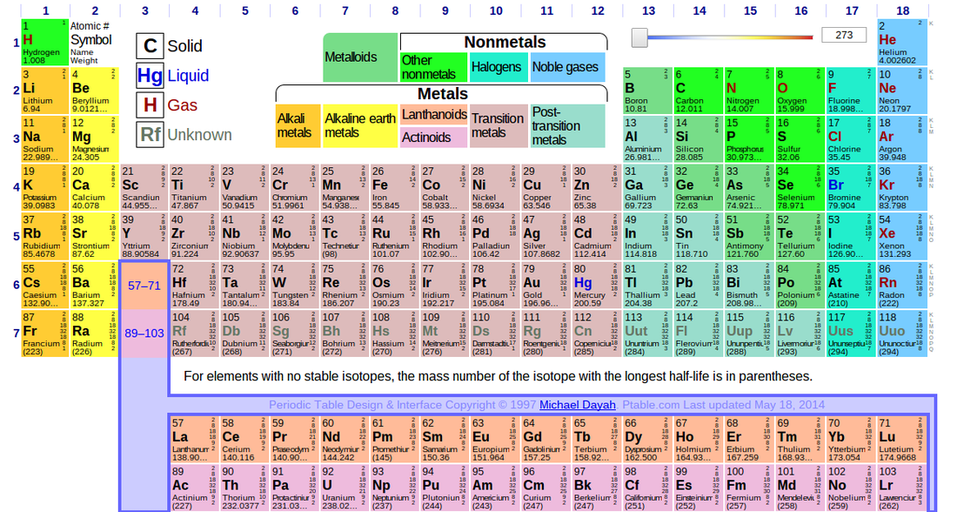
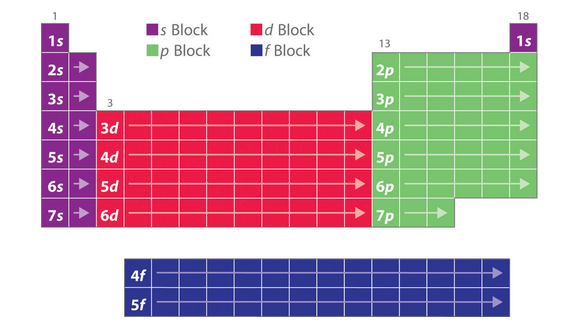
How to measure an atom?
- The electron cloud does not have a definite edge.
- Atomic radius- this is half the distance between the two nuclei of a diatomic molecule.
Diatomic Molecules
- There is seven diatomic molecules
- The diatomic molecules always come in pairs
- H2 , N2 , O2 , F2 , Cl2 , Br2 , I2 are the seven diatomic molecules
What is an ion?
- Some compounds are made up of particles called ions.
- Ion- an atom (or group of atoms ) that has a positive or negative charge.
- Atoms are normally neutral because the amount of protons matches the amount of electrons
- Ions are made when the atoms are no longer neutral. This means the atoms have a positive or negative charge.
- Metals tend to lose electrons from the outermost energy level.
- Sodium losses one : now has more protons (11) than electrons(10) thus has a positive charge=cations
- Nonmetals tend to gain one or more electrons.
- Chlorine gains an electron
- Protons(17) no longer equals the electrons(18), so charge of -1
- Chlorine is now called Chloride-ion
What is a trend?
- Periodic trends are influenced by 2 primary factors.
- Energy level: higher the energy level the further the electrons are from the nucleus.
- If you add more protons it can be a change to the nucleus because the electrons will be pulled in tighter to the nucleus.
- The electron cloud does not have a definite edge.
- Atomic radius- this is half the distance between the two nuclei of a diatomic molecule.
Diatomic Molecules
- There is seven diatomic molecules
- The diatomic molecules always come in pairs
- H2 , N2 , O2 , F2 , Cl2 , Br2 , I2 are the seven diatomic molecules
What is an ion?
- Some compounds are made up of particles called ions.
- Ion- an atom (or group of atoms ) that has a positive or negative charge.
- Atoms are normally neutral because the amount of protons matches the amount of electrons
- Ions are made when the atoms are no longer neutral. This means the atoms have a positive or negative charge.
- Metals tend to lose electrons from the outermost energy level.
- Sodium losses one : now has more protons (11) than electrons(10) thus has a positive charge=cations
- Nonmetals tend to gain one or more electrons.
- Chlorine gains an electron
- Protons(17) no longer equals the electrons(18), so charge of -1
- Chlorine is now called Chloride-ion
What is a trend?
- Periodic trends are influenced by 2 primary factors.
- Energy level: higher the energy level the further the electrons are from the nucleus.
- If you add more protons it can be a change to the nucleus because the electrons will be pulled in tighter to the nucleus.
Citations:
- Dayah, Michael. "Dynamic Periodic Table." Dynamic Periodic Table. N.p., n.d. Web. 28 May 2014.
- Boxeman Science. "Periodicity." YouTube. YouTube, 05 Aug. 2013. Web. 29 May 2014.
- "Building Up the Periodic Table." Building Up the Periodic Table. N.p., n.d. Web. 29 May 2014.